Research Introduction
Development of Biocatalysts Based on Cytochrome P450s
Chapter 2 Functional modulation of P450BM3 using decoy molecule and its application to biocatalyst
Episode 11: Expansion of applicability of decoy molecules in whole-cell reactions in Escherichia coli by designing outer membrane proteins
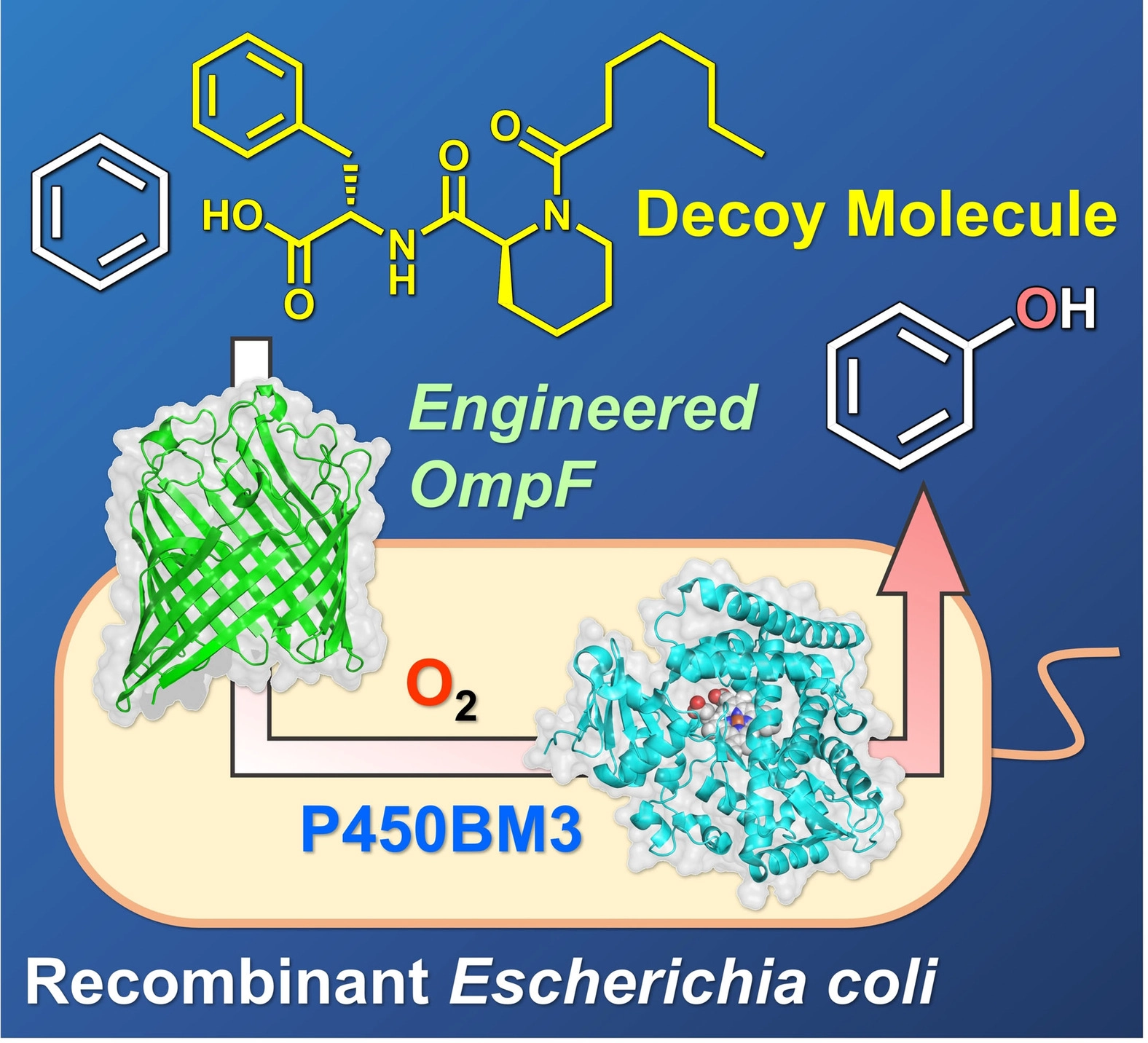
We previously succeeded in the direct hydroxylation of benzene to phenol by adding decoy molecules to P450BM3 overexpressed in Escherichia coli. This method does not require enzyme expression or purification procedures compared with conventional reaction systems using purified P450BM3. In addition, the coenzyme NADPH, which requires a stoichiometric amount for the reaction to proceed, is regenerated by the metabolic system within E. coli, making this an excellent reaction system with many advantages, such as not requiring the external addition of NADPH. However, the decoy molecules added externally must penetrate the outer and inner membranes of E. coli to reach P450BM3 in the cytoplasm, and applicable decoy molecules were limited to zwitterionic molecules such as C7-Pro-Phe. Most decoy molecules that are effective against purified enzymes do not have zwitterionic properties and are, therefore, ineffective in reactions within E. coli.
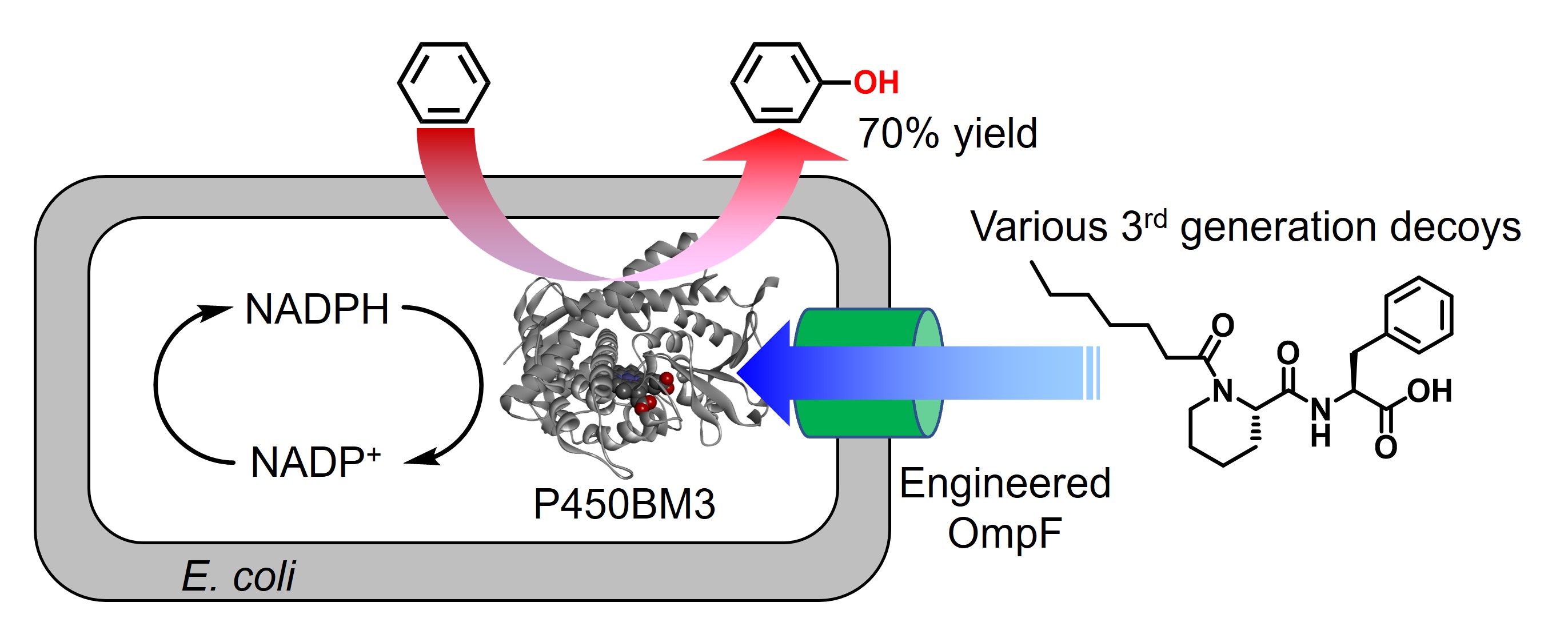
To identify proteins involved in the uptake of C7-Pro-Phe through the outer membrane, we focused on the outer membrane proteins OmpC and OmpF. These are outer membrane proteins with a barrel-like structure that non-selectively uptake small hydrophilic molecules, including antibacterial agents. We knocked out OmpR, which is responsible for the transcriptional control of OmpC and OmpF, to prepare E. coli that does not express both OmpC and OmpF, and then transformed this with plasmids encoding either OmpC or OmpF, respectively, to prepare E. coli that expressed OmpC or OmpF alone. When C7-Pro-Phe was added to these E. coli and benzene hydroxylation was performed, it was found that OmpF-expressing E. coli had a higher benzene hydroxylation efficiency than OmpC-expressing E. coli, indicating that OmpF was predominant in the uptake of C7-Pro-Phe. In addition, based on the crystal structure of OmpF, we designed a mutant OmpFΔ108-130 in which the loop that narrows the space inside the barrel was removed. When they expressed this mutant in E. coli, we found that the benzene hydroxylation efficiency was improved for various decoy molecules that had previously been difficult to take up by E. coli. Furthermore, OmpFΔ108-130 promoted the uptake of not only decoy molecules but also substrates such as benzene, significantly improving the efficiency of reactions within E. coli. In particular, C7AM-Pip-Phe, which showed excellent activity in benzene hydroxylation in purified P450BM3 enzyme, gave a phenol yield of 5.7% in normal E. coli, because of low efficiency of uptake of the decoy molecule into E. coli. however, in E. coli expressing OmpFΔ108-130, the phenol yield was improved to 70%. Furthermore, E. coli expressing OmpFΔ108-130 was also effective in hydroxylating unnatural substrates other than benzene and could carry out whole-cell reactions while maintaining a selectivity similar to that of the purified enzyme.
Please refer to this paper for details.
- M. Karasawa, K. Yonemura, J. K. Stanfield, K. Suzuki, O. Shoji "Designer Outer Membrane Protein Facilitates Uptake of Decoy Molecules into a Cytochrome P450BM3-Based Whole-Cell Biocatalyst (English ver.)" "Ein Designeraußenmembranprotein fördert die Aufnahme von Täuschmolekülen in einen auf Zytochrom P450BM3 beruhenden Ganzzellbiokatalysator (German ver.)" , Angew. Chem. Int. Ed., 61, (2022) e202111612. Angew. Chem., 134, (2022) e202111612.
https://doi.org/10.1002/anie.202111612
https://doi.org/10.1002/ange.202111612